
Know all the risks
Most people decided to take the injection blindly, without having all the cards in hands. More than ever, when facing the choice about the current Cov-vaccination, informed consent is paramount. Knowing all the risks is one key element of being informed.
Is there reasons for concerns?
Vaccine adverse events in the new millenium; is the reason for concern?
Bulletin of the World Health Organization 2000; 78(2): 205–215. B. J. Ward
ABSTRACT
As more and more infectious agents become targets for immunization programmes, the spectrum of adverse events linked to vaccines has been widening. Although some of these links are tenuous, relatively little is known about the immunopathogenesis of even the best characterized vaccine-associated adverse events (VAAEs). The range of possible use of active immunization is rapidly expanding to include vaccines against infectious diseases that require cellular responses to provide protection (e.g. tuberculosis, herpes viral infections), therapeutic vaccines for chronic infections (e.g. human immunodeficiency virus (HIV) infection, viral hepatitis B and C), and vaccines against non-infectious conditions (e.g. cancer, autoimmune diseases). Less virulent pathogens (e.g. varicella, rotavirus in the developed world) are also beginning to be targeted, and vaccine use is being justified in terms of societal and parental “costs” rather than in straightforward morbidity and mortality costs. In the developed world the paediatric immunization schedule is becoming crowded, with pressure to administer increasing numbers of antigens simultaneously in ever simpler forms (e.g. subcomponent, peptide, and DNA vaccines). This trend, while attractive in many ways, brings hypothetical risks (e.g. genetic restriction, narrowed shield of protection, and loss of randomness), which will need to be evaluated and monitored. The available epidemiological and laboratory tools to address the issues outlined above are somewhat limited. As immunological and genetic tools improve in the years ahead, it is likely that we shall be able to explain the immunopathogenesis of many VAAEs and perhaps even anticipate and avoid some of them. However, this will only happen if the human and financial resources needed for monitoring and studying vaccine safety stay in step with the accelerating pace of vaccine development. Failure to make such a commitment would put all immunization programmes at risk.
Are they safe and effective?
The fact that the current Cov-vaccine candidates were rushed into market before going through the regular channel of safety is concerning. We are witnessing parenteral and societal cost overriding the human cost. These previously unjustified costs are now dangerously being considered in the cost benefit analysis of vaccination rollout. It is wise to question this conclusion ‘’the benefit outweighs the risk’’. How much does a third party (government and industry) has outside interests in addition to the health of the intended vaccinees in this Cov-vaccine rollout? Especially when the individual is not particularly affected by the disease (healthy children and young adults). Systemic and mandated vaccination may not benefit the individual in this instance. Therefore it is your duty, as an individual (future inoculee) to know all the risk related to the current Cov-vaccination. Review clinical trial data, visit different adverse effects databases and listen to testimonials of inoculation injured individuals. Performing your own due diligence, you can see for yourself if those emergency use approved vaccine candidates are safe and effective.
Review of clinical trials
As seen in pervious lessons, clinical trials are one of the key element of the safety net established to ensure population safety against any drug for vaccine set to go on market. Ordinarily, trust is given to many entity to review and give oversight on these clinical trial data. We step on dangerous grounds when, as mentioned in the previous WOH bulletin, outside interest may compete with the individual interest. The controversy and divide observed around the different SARS-CoV-2 vaccine candidate can be reflections of the competing interests. Can you still blindly rely on the regulatory agency to defend your interest?
We cannot afford to wait and see, as decisions towards inoculations need to be taken by each individual either for themselves or their dependents. Every one need to be able to get to the data, perform their review and analysis and come up with unbiases risk- benefit conclusion. It is unrealistic to believe the the general population has the ability to do so. Due to the importance of the matter, Minority Check scientific team made the process accessible.

COVID-19 Vaccine candidate AstraZeneca
COVID-19 Vaccine AstraZeneca
Common name: COVID-19 Vaccine (ChAdOx1-S [recombinant])
Pfizer Clinical Trial
Post marketing adverse events – Including a weekly (Friday) VAERS update
We can also learn from other people’s experiences. Clinical trials are mainly a glimpse of what can happen when the product is rolled out into market. Especially for these clinical trials which are not even fully completed. Therefore, different sources of information are needed to have the most accurate image of the reality. Although adverse event reporting amount to 1-2% of reality occurrence, it is worthy of acknowledgment as an important part of the process of being fully informed.
VAERS
| Astra-Zeneca: AZD1222 | Serious adverse events * | Death* |
| JANSSEN: JNJ-78436735 | Serious adverse events | Death |
| Moderna: mRNA-1273 | Serious adverse events | Death |
| Pfizer-BioNTech: TOZINAMERAN, Comirnaty | Serious adverse events | Death |
*Astra-Zeneca data can no longer be found in the VAERS database, their emergency approved vaccine candidate was removed from market after a few months of being administered. You will be redirected to the EU database for adverse events
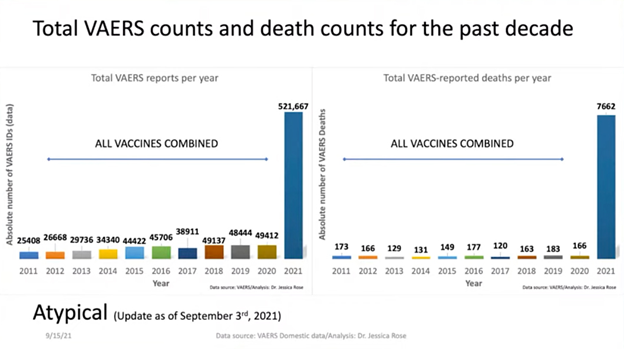
The treacherous aspect of the different inoculations against SARS-CoV-2 virus is well observed by their aftermath resulting in a near 1000% increase of events and death counts in 2021 compared to the combined data from the past decade ( 2011-2020).
The Norwegian Medicines Agency
The Norwegian Medicines Agency publishes overviews of suspected adverse reactions associated with corona vaccination in Norway. The overviews are usually published every Thursday or Friday.
Status as of September 21:
- Number of persons vaccinated with first dose: 4,092,840
- Number of persons vaccinated with second dose: 3,570,608
- Reported suspected adverse reactions: 33,287
- Assessed suspected adverse reactions: 18,054
EU looking into new possible side-effects of mRNA COVID-19 shots
Pushkala AripakaAugust 11, 2021·2 min read
By Pushkala Aripaka
(Reuters) -Three new conditions reported by a small number of people after vaccination with COVID-19 shots from Pfizer and Moderna are being studied to assess if they may be possible side-effects, Europe’s drugs regulator said on Wednesday.
Erythema multiforme, a form of allergic skin reaction; glomerulonephritis or kidney inflammation; and nephrotic syndrome, a renal disorder characterised by heavy urinary protein losses, are being studied https://www.ema.europa.eu/en/documents/covid-19-vaccine-safety-update/covid-19-vaccine-safety-update-spikevax-previously-covid-19-vaccine-moderna-11-august-2021_en.pdf by the safety committee of the European Medicines Agency (EMA), according to the regulator https://www.ema.europa.eu/en/documents/covid-19-vaccine-safety-update/covid-19-vaccine-safety-update-comirnaty-11-august-2021_en.pdf.
Pfizer, by far the biggest supplier of COVID-19 vaccines to the European Union, and Moderna did not immediately respond to Reuters requests for comment.
Just over 43.5 million doses of Moderna’s vaccine, Spikevax, have been administered in the European Economic Area as of July 29, the EMA said, compared to more than 330 million doses of the Pfizer shot, Comirnaty, developed with Germany’s BioNTech.
Last month, the EMA found a possible link https://www.reuters.com/business/healthcare-pharmaceuticals/eu-regulator-lists-heart-condition-possible-side-effect-mrna-vaccines-2021-07-09 between very rare heart inflammation and the mRNA vaccines. However, the European regulator and the World Health Organization have stressed that benefits from these vaccines outweigh any risks.
The watchdog did not give details on Wednesday on how many cases of the new conditions were recorded, but said it had requested more data from the companies. The EMA did not recommend changes to the labelling of the vaccines.
It disclosed the new assessments as part of routine updates to the safety section of all authorised vaccines’ database and added menstrual disorders as a condition it was studying for vaccines, including those from AstraZeneca and J&J, after the EMA’s update https://www.reuters.com/business/healthcare-pharmaceuticals/eu-regulator-lists-heart-condition-possible-side-effect-mrna-vaccines-2021-07-09 last week.
Moderna’s shares, which have climbed more than 75% to Tuesday’s close since mid-July when it joined the S&P 500 index, fell about 16% to $384 on Wednesday.
BioNTech’s U.S.-listed shares were down 14.4% and Pfizer nearly 4%.
(Reporting by Pushkala Aripaka in Bengaluru; additional reporting by Manojna Maddipatla; Editing by Shounak Dasgupta and Mark Potter)
Take away
Contrary to the SARS-CoV-2 virus, the victimology of the vaccine has not yet been assessed nor addressed. With the virus, it is now well understood who stand to be impacted by serious disease or death. On the other hand, such affirmation cannot be made toward the various emergency approved vaccine candidates. The reasons why some people encounter serious adverse events or death after inoculation are still mystery. Therefore, regardless of the degree of incidence (rare or not), for the inoculees, this amounts to playing Russian roulette. Additionally, the inoculation injured individuals are desperately left to fend for themselves, as they struggle to be recognized and as treatment for some of the most serious neurological disorders are either non-existent or unknown.
Facing vaccine injuries
Mass media is doing an excellent job using their platform to display solely the impact of SARS-CoV-2 infection. The only give voice to the victims of the virus, maintaining the population in a fearful environment. But those who became victims of the injection also deserve attention since they gave their arm to tame their acquired fear, to do the right thing, to help society, to go along with societal expectations.
SARS-CoV-2 inoculation injuries are not some urban legends. Real people are dealing with them everyday; they must not be ignored and their ordeal must not taken lightly. They deserve attention to prevent future tragic outcomes (to individuals health or societal safety from fast arising dangerous divide).
Take the necessary time to see them, to listen to them and to feel compassion for them. Understand those who don’t want to take the same risk. Understand that this easily could have been you.
Testimonials
More testimonials

Compassion
One of the main objectives of the Cov-Awareness vs Cov-friction course is to instill a strong dose of compassion into each of us, since it seems like this feeling is slowly leaving our life environment. Through knowledge of one’s experience, perhaps understanding of one another will happen and compassion will follow suit. Saving not only the ever so important medical freedom, saving what humanity stands for: connection to each other.
The world is witnessing more and more instances of harshness, unkindness, judgment towards people closest to oneself, simply because of some differences of opinion. Compassion seems to be felt only when the issue or the opponent is remote, far from the individual. Compassion takes the exit route, even more so when fear comes into play. Coronaphobia has become so powerful, it is leading its subjects to sentence anyone (mainly the not cov-vaccinated) who don’t cater to the same fear, to pitiless discrimination and breach of their freedom. Where is this going to lead to? Nowhere good…Fast.
